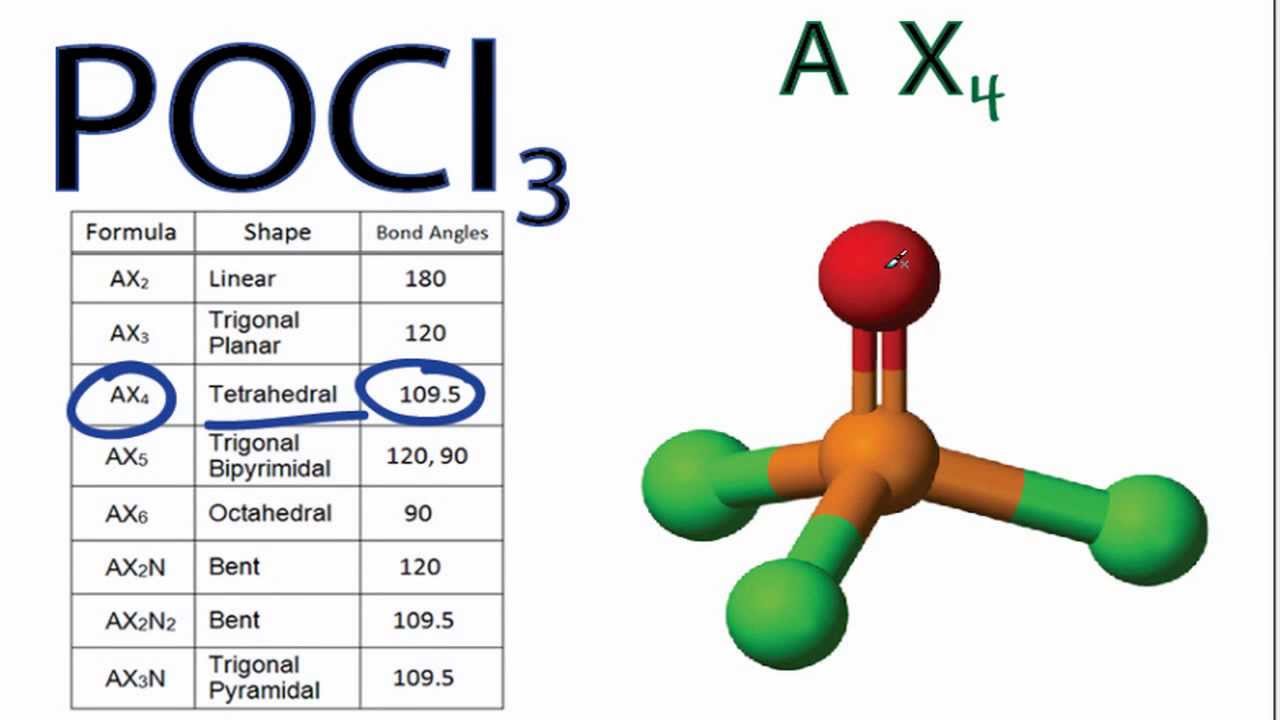
POCl3 Lewis structure, molecular geometry or shape, electron geometry, bond angle, formal charges | Molecular geometry, Molecular, Electron configuration

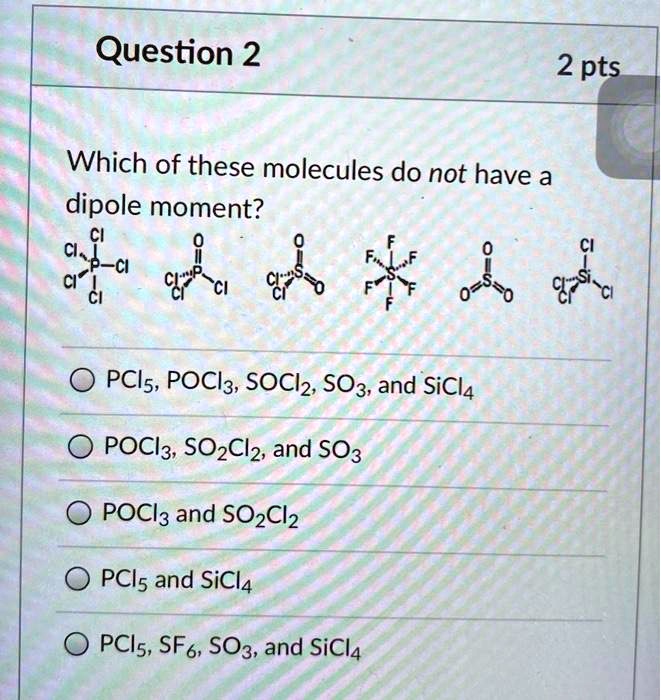
Welcome to Chem Zipper.com......: Among I2, ICI, CCl4, PCl3, POCl3, which molecules exhibit dipole moment?

SOLVED: Answer the following question: Draw a Lewis structure for the compound phosphoryl chloride (POCl3). Mention the type of bond in the compound. Is the compound POCl3 polar or nonpolar?

SOLVED: Answer the following question: Draw a Lewis structure for the compound phosphoryl chloride (POCl3). Mention the type of bond in the compound. Is the compound POCl3 polar or nonpolar?